Research
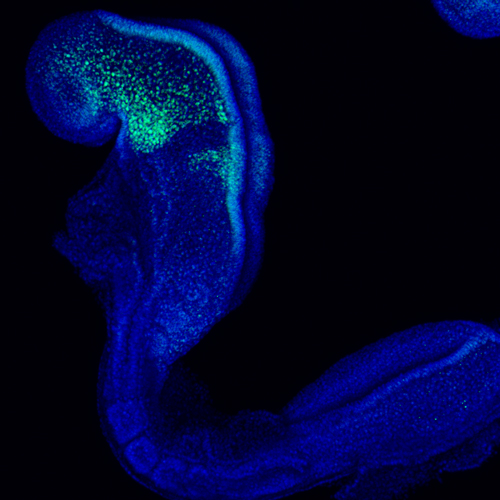
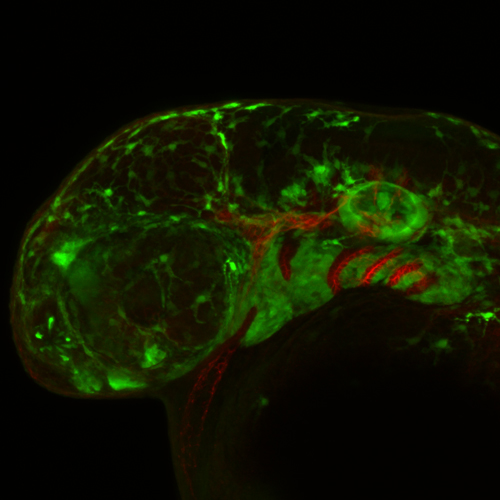
Congenital anomalies occur in 1% of all live human births. Approximately one third of these developmental anomalies affect craniofacial development, which depends upon multipotent neural crest cells. Neural crest cells are induced in the neuroepithelium and migrate throughout the body where they differentiate into a variety of tissues such as craniofacial bone, cartilage and connective tissue, as well as neurons and glia of the peripheral nervous system and pigment cells in the skin. Depending on the stage and axial level of the disruption in neural crest cell development, distinct phenotypes of the craniofacial, cardiac or enteric nervous system tissue occur. Therefore, it’s important to understand the mechanisms that regulate neural crest cell development.
My research in the Trainor Lab is focused on understanding the tissue specific roles of factors required in global transcription and translation in development and disease.
My research in the Trainor Lab is focused on understanding the tissue specific roles of factors required in global transcription and translation in development and disease.
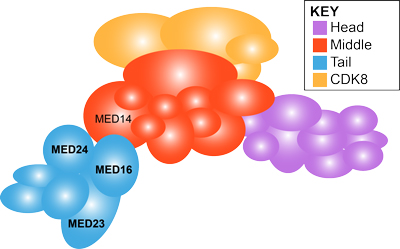
Mediator complex and craniofacial development
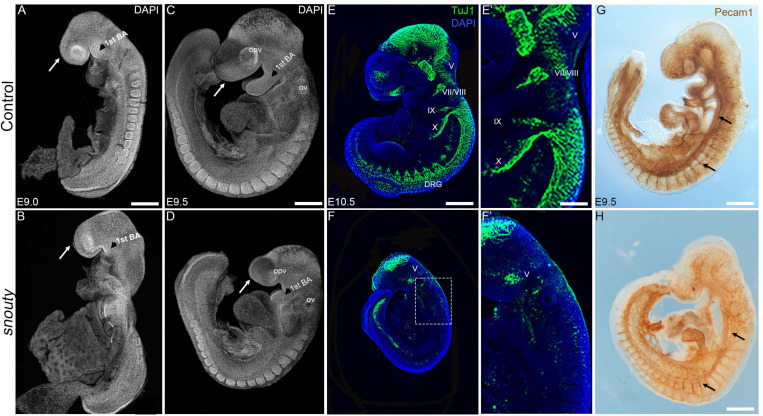
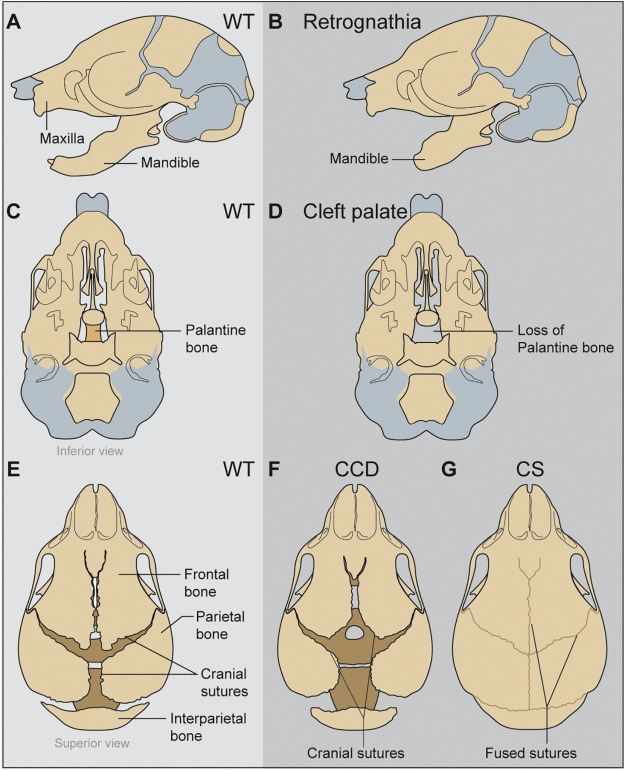
The Mediator complex is a multiprotein complex that forms a molecular bridge between RNA polymerase II and transcription factors and thus is required in all cells for RNA polymerase II driven transcription. I have observed that a point mutation in a tail Mediator complex subunit Med23results in severe craniofacial malformations that arise from defects in neural crest development. This raises the question of how disruptions in a ubiquitously expressed gene and a global process results in tissue specific defects. To understand if Med23 has a cell-autonomous function in neural crest cells, I generated neural crest cell specific deletion mutants of Med23 and observed micrognathia, glossoptosis and cleft palate reminiscent of Pierre-Robin Sequence syndrome. On further molecular inspection, I have uncovered a new function for Med23 by demonstrating that in neural crest cell derived mesenchyme, Med23 binds directly to the promoters and enhancers of genes, and regulates their expression and downstream targets required for craniofacial cartilage and bone development. These findings demonstrate a link between the global transcriptional machinery and developmental signaling pathways that elicit tissue specific effects in development and disease. My future research is focused on parsing the function of individual Mediator complex subunits in craniofacial development and congenital disorders.
Funded by K99 NIDCR
Funded by K99 NIDCR
RNA Polymerase I and craniofacial development
Ribosome biogenesis is a global process required for protein synthesis in all cells, yet disruptions result in tissue specific defects, which are known as ribosomopathies. Treacher Collins Syndrome (TCS) is one such ribosomopathy that arises from mutations in the RNA Polymerase I (Pol I)subunits POLR1C and POLR1D as well as in a Pol I associated factor, TCOF1. TCS is characterized by craniofacial anomalies such as underdevelopment of the zygomatic complex, cheekbones, jaws, palate and mouth resulting in difficulty breathing and feeding. In collaboration with members of the Trainor lab, I aim to identify the molecular mechanisms underlying the etiology of TCS by characterizing mouse animal and embryonic fibroblast cell derived models of TCS.
In addition to the craniofacial anomalies characteristic of TCS, conditional knockout of Pol I subunits and Tcof1 in neural crest cells also results in total intestinal aganglionosis, the most severe form of Hirschsprung disease. Hirschsprung disease is characterized by the absence of ganglia from variable regions of the gastrointestinal tract and is caused by incomplete colonization of the gut by neural crest cells and their failure to form a complete enteric nervous system. Therefore, I am investigating the function of Pol I subunits and Tcof1 in enteric nervous system development and in the etiology of Hirschsprung disease. This work has linked global RNA Polymerase I transcription to tissue specific neurocristopathies affecting the craniofacial skeleton and gastrointestinal system.
My future research aims to understand the molecular function of Pol I associated factors in the context of vertebrate development and birth defects.
Funded by AAA
In addition to the craniofacial anomalies characteristic of TCS, conditional knockout of Pol I subunits and Tcof1 in neural crest cells also results in total intestinal aganglionosis, the most severe form of Hirschsprung disease. Hirschsprung disease is characterized by the absence of ganglia from variable regions of the gastrointestinal tract and is caused by incomplete colonization of the gut by neural crest cells and their failure to form a complete enteric nervous system. Therefore, I am investigating the function of Pol I subunits and Tcof1 in enteric nervous system development and in the etiology of Hirschsprung disease. This work has linked global RNA Polymerase I transcription to tissue specific neurocristopathies affecting the craniofacial skeleton and gastrointestinal system.
My future research aims to understand the molecular function of Pol I associated factors in the context of vertebrate development and birth defects.
Funded by AAA
Teaching and Service
Teaching experience
Stowers Institute for Medical Research
November 2019 & November 2020
Lecture on Organogenesis for Cellular Dynamics and Developmental Biology Module for Graduate School at Stowers Institute for Medical Research
Marine Biology Laboratory
June 2018 & June 2019
Teaching assistant, Mouse embryology, Embryology course
University of Delaware, Newark, DE
February 2013 – December 2017
Teaching assistant (TA), Introductory Biology I & II
Mentoring experience
Carlee Roberts
Spring 2021
KU Med Practicum Student at Stowers Institute for Medical Research
Spring 2021
KU Med Practicum Student at Stowers Institute for Medical Research
Padmini Sreedhara
Summer 2019
Undergraduate summer intern at Stowers Institute for Medical Research
Summer 2019
Undergraduate summer intern at Stowers Institute for Medical Research
Nathaniel Borders
Fall 2014 - Spring 2017
Undergraduate senior thesis titled Functional characterization of RNA binding protein Caprin 2 in mouse eye development. Current placement: Research Assistant in Dr. George Church's lab at Harvard University.
Fall 2014 - Spring 2017
Undergraduate senior thesis titled Functional characterization of RNA binding protein Caprin 2 in mouse eye development. Current placement: Research Assistant in Dr. George Church's lab at Harvard University.
Aishwarya Krishnakumar
Summer 2014
Professional Science Masters student for a summer volunteer program
Summer 2014
Professional Science Masters student for a summer volunteer program
Committees
2022
Scientific affairs committee
American Association for Anatomy
American Association for Anatomy
2021
DEI council
Stowers Institute for Medical Research
Stowers Institute for Medical Research
2015 - 2017
Safety committee
Department of Biological Sciences
University of Delaware
Department of Biological Sciences
University of Delaware
2016 - 2017
TA training fellow for the Center for Teaching & Assessment of Learning’s “Teaching Assistant Orientation”
University of Delaware
University of Delaware
2016
Teaching and Technology Committee
Dept. of Biological Sciences
University of Delaware
Dept. of Biological Sciences
University of Delaware
2015
Vice President of Biological Graduate Students Association (BGSA)
University of Delaware
University of Delaware
2014
Secretary of Biological Graduate Students Association (BGSA)
University of Delaware
University of Delaware
Funding, Honors and Awards
Funding
2022
K99/R00 - Project Number K99DE030972
Functional Characterization of Mediator Complex Proteins in Neural Crest and Craniofacial Development
Functional Characterization of Mediator Complex Proteins in Neural Crest and Craniofacial Development
2021
American Association for Anatomy Post-Doctoral Fellowship
This award provides salary support to postdoctoral trainees working in any aspect of biology relevant to the anatomical sciences, including both basic science research and education research.
This award provides salary support to postdoctoral trainees working in any aspect of biology relevant to the anatomical sciences, including both basic science research and education research.
2016
Earl & Shirley Herbst eye research award for Summer Student Fellowship from Fight for Sight, Inc.
FFS Fellowships were competitively awarded to top 14 students in eye research nationwide in 2015, and only one was selected for the Earl & Shirley Herbst Eye Research Foundation Award
FFS Fellowships were competitively awarded to top 14 students in eye research nationwide in 2015, and only one was selected for the Earl & Shirley Herbst Eye Research Foundation Award
2016
University Graduate Fellowship Award, University of Delaware
This award is given to 1 PhD student among ~80 in recognition of academic achievements and potential for success in the field of study.
This award is given to 1 PhD student among ~80 in recognition of academic achievements and potential for success in the field of study.
Awards
2021
Second place in Postdoctoral Poster Presentation Competition
44th Annual Meeting of the Society of Craniofacial Genetics and Developmental Biology
44th Annual Meeting of the Society of Craniofacial Genetics and Developmental Biology
2021
First place in Postdoctoral Poster Presentation Competition,
North-East Society of Developmental Biology Meeting
North-East Society of Developmental Biology Meeting
2020
Third place in Postdoctoral Poster Presentation Competition
43rd Annual Meeting of the Society of Craniofacial Genetics and Developmental Biology
43rd Annual Meeting of the Society of Craniofacial Genetics and Developmental Biology
2019
Best Trainee Presentation Award
42nd Annual Meeting of the Society of Craniofacial Genetics and Developmental Biology
42nd Annual Meeting of the Society of Craniofacial Genetics and Developmental Biology
2019
Travel Grant from Society of Development Biology to attend 42nd Annual Meeting of the Society of Craniofacial Genetics and Developmental Biology
2017
Travel Grant from Society of Development Biology to attend 76th Annual Meeting of the Society of Developmental Biology
2017
Travel Grant Award
Department of Biological Sciences
University of Delaware
Department of Biological Sciences
University of Delaware
2016
Excellent Graduate Teaching Assistant Award in Biological Sciences
University of Delaware
This annual award is given to 2 Teaching assistants each year among ~40 in recognition of excellence in teaching
University of Delaware
This annual award is given to 2 Teaching assistants each year among ~40 in recognition of excellence in teaching
2016
Professional Development Award
Office of Graduate Studies
University of Delaware
Office of Graduate Studies
University of Delaware
2016
Travel Grant from Society of Development Biology to attend 75th Annual Meeting of the Society of Developmental Biology
2016
Travel Grant Award
Department of Biological Sciences
University of Delaware
Department of Biological Sciences
University of Delaware
2015
Knight Templers Travel Grant to attend the Annual Meeting of the Association for Research in Vision and Ophthalmology (ARVO) in 2015
Competitively awarded to select few trainees with top scoring abstracts at ARVO
Competitively awarded to select few trainees with top scoring abstracts at ARVO
2014
Members-in-training (MIT) Outstanding Poster Award, Lens Section
Association of Research in Vision and Ophthalmology (ARVO) Annual Meeting.
This international award is given to top 1 lens trainee researcher from ~200 abstracts considered. I won this highly competitive award as a graduate student among all lens research trainees (both post-doctoral and graduate trainees compete in the same group)
Association of Research in Vision and Ophthalmology (ARVO) Annual Meeting.
This international award is given to top 1 lens trainee researcher from ~200 abstracts considered. I won this highly competitive award as a graduate student among all lens research trainees (both post-doctoral and graduate trainees compete in the same group)
2014
Professional Development Award
Office of Graduate Studies
University of Delaware
Office of Graduate Studies
University of Delaware
2014
Travel Grant Award
Department of Biological Sciences
University of Delaware
Department of Biological Sciences
University of Delaware
Publications
2022
RNA transcription is integral to liquid-liquid phase separation and maintenance of nucleolar structure
Soma Dash, Maureen C. Lamb, Jeffrey J Lange, Mary Cathleen McKinney, Dai Tsuchiya, Fengli Guo, Xia Zhao, Timothy J Corbin, MaryEllen Kirkman, Kym Delventhal, Emma L Moore, Sean A McKinney, Rita Shiang, Paul Trainor. bioRxiv 2022.11.14.516489.
Soma Dash, Maureen C. Lamb, Jeffrey J Lange, Mary Cathleen McKinney, Dai Tsuchiya, Fengli Guo, Xia Zhao, Timothy J Corbin, MaryEllen Kirkman, Kym Delventhal, Emma L Moore, Sean A McKinney, Rita Shiang, Paul Trainor. bioRxiv 2022.11.14.516489.
Dynamic regulation and requirement for ribosomal RNA transcription during mammalian development
Falcon KT, Watt KEN, Dash S, Zhao R, Sakai D, Moore EL, Fitriasari S, Childers M, Sardiu ME, Swanson S, Tsuchiya D, Unruh J, Bugarinovic G, Li L, Shiang R, Achilleos A, Dixon J, Dixon MJ, Trainor PA. Proc Natl Acad Sci U S A. 2022;119:e2116974119 doi: 10.1073/pnas.2116974119.
Falcon KT, Watt KEN, Dash S, Zhao R, Sakai D, Moore EL, Fitriasari S, Childers M, Sardiu ME, Swanson S, Tsuchiya D, Unruh J, Bugarinovic G, Li L, Shiang R, Achilleos A, Dixon J, Dixon MJ, Trainor PA. Proc Natl Acad Sci U S A. 2022;119:e2116974119 doi: 10.1073/pnas.2116974119.
Nucleolin loss-of-function leads to aberrant FGF signaling and craniofacial anomalies.
Dash S, Trainor PA. [published ahead of print June 29 2022]. Development. 2022;149.
Dash S, Trainor PA. [published ahead of print June 29 2022]. Development. 2022;149.
2021
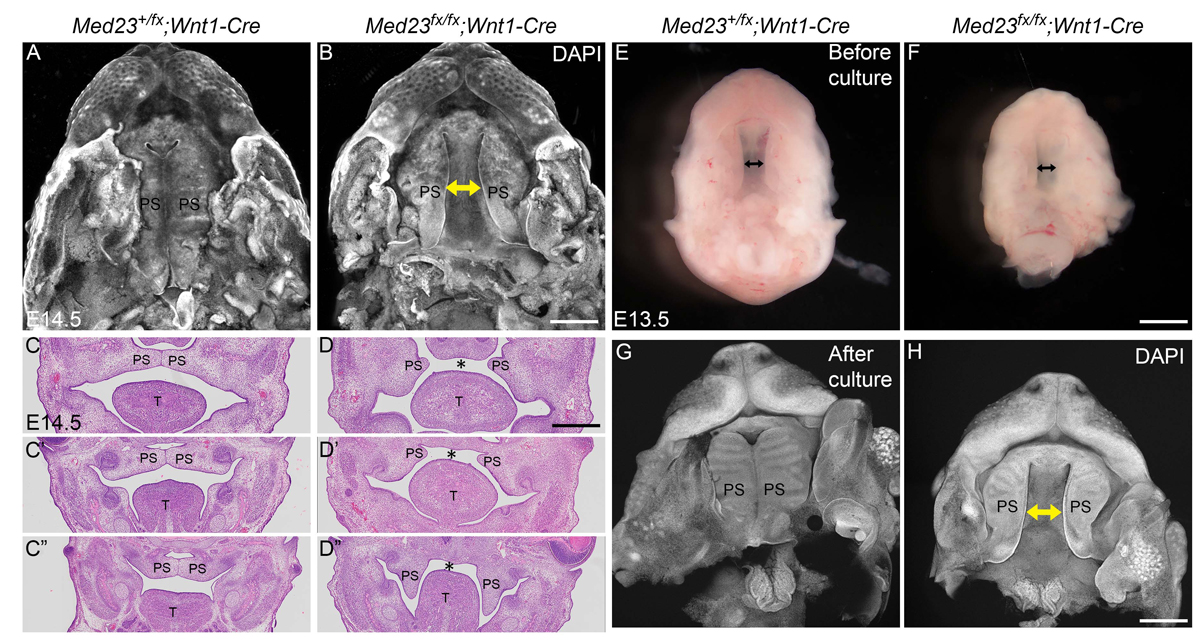
Med23 Regulates Sox9 Expression during Craniofacial Development
Dash S, Bhatt S, Falcon KT, Sandell LL, Trainor PA. J Dent Res. 2021;100:406-414.
Dash S, Bhatt S, Falcon KT, Sandell LL, Trainor PA. J Dent Res. 2021;100:406-414.
2020
The Mediator subunit, Med23 is required for embryonic survival and regulation of canonical WNT signaling during cranial ganglia development
Dash S, Bhatt S, Sandell L, Seidel C, Ahn Y, Krumlauf R, Trainor P. Front Physiol. 2020;11:531933. doi: 531910.533389/fphys.532020.531933.
Dash S, Bhatt S, Sandell L, Seidel C, Ahn Y, Krumlauf R, Trainor P. Front Physiol. 2020;11:531933. doi: 531910.533389/fphys.532020.531933.
The development, patterning and evolution of neural crest cell differentiation into cartilage and bone
Dash S, Trainor P. [published online ahead of print May 14 2020]. Bone. 2020;115409. doi:10.1016/j.bone.2020.115409.
Dash S, Trainor P. [published online ahead of print May 14 2020]. Bone. 2020;115409. doi:10.1016/j.bone.2020.115409.
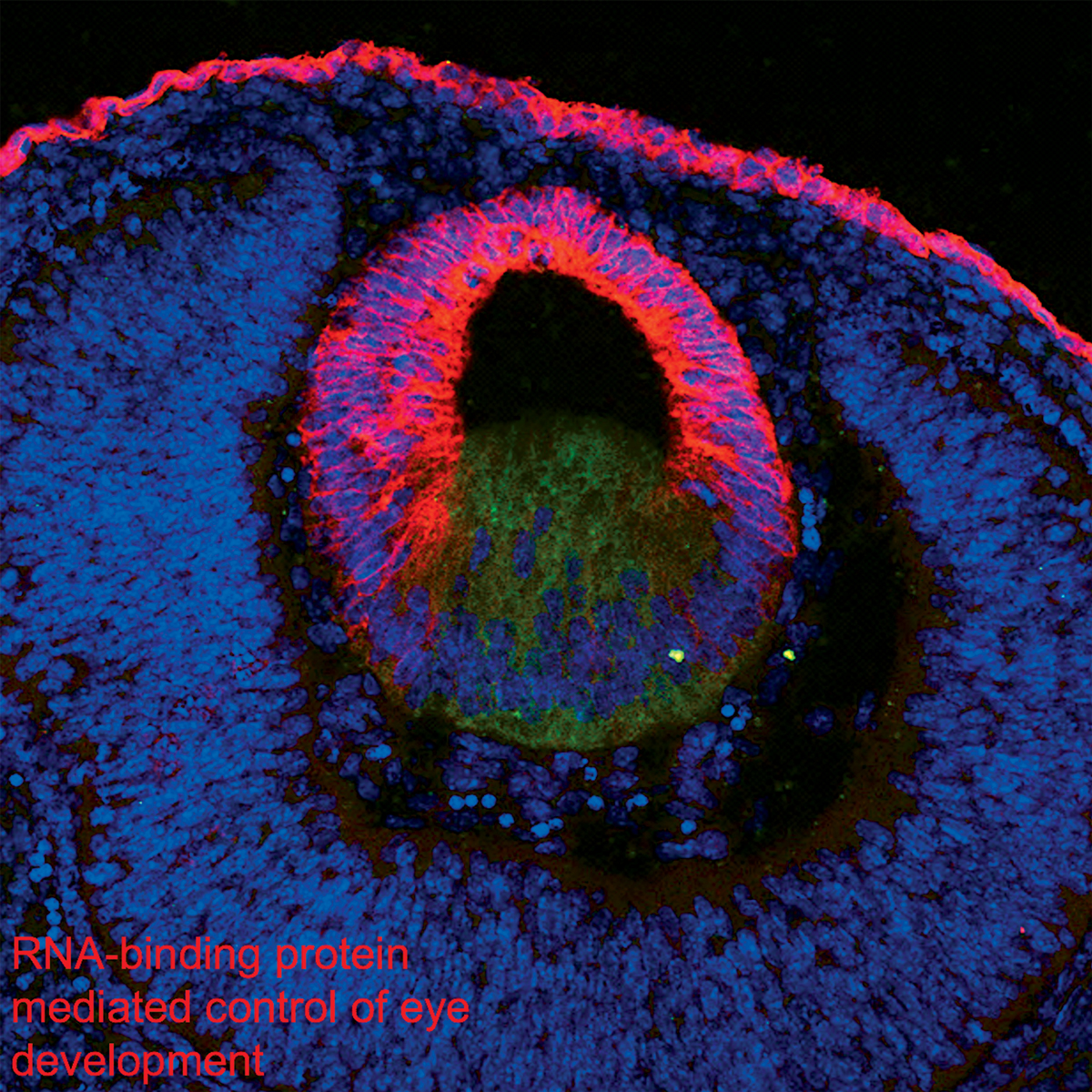
The master transcription factor SOX2, mutated inanophthalmia/microphthalmia, is post-transcriptionally regulated by the conserved RNA-binding protein RBM24 in vertebrate eye development.
Dash S, Brastorm LK, Patel SD, Scott CA, Slusarksi D C, Lachke SA Human Molecular Genetics. (2020)
Dash S, Brastorm LK, Patel SD, Scott CA, Slusarksi D C, Lachke SA Human Molecular Genetics. (2020)
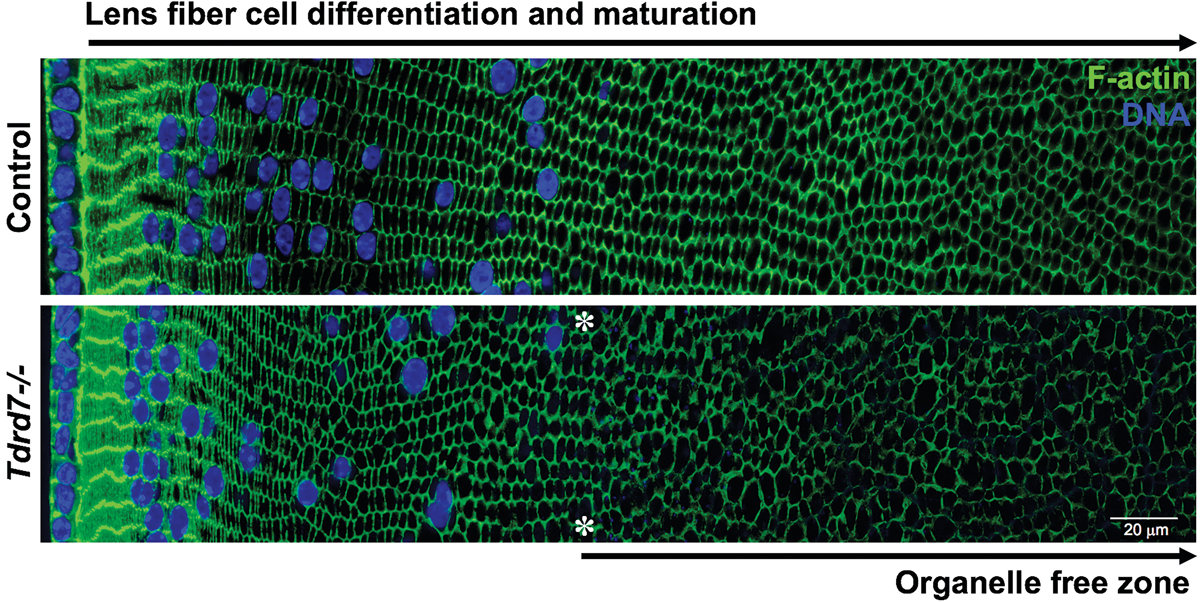
The Tudor-domain protein TDRD7, mutated in congenital cataract, controls the heat shock protein HSPB1 (HSP27) and lens fiber cell morphology
Barnum C.E., Saai S.A., Patel S.D., Cheng C., Anand D., Xu X., Dash S., Siddam A.D., Glazewski L., Paglione E., Polson S., Chuma S., Mason R.W., Wei, S., Batish M., Fowler V.M., Lachke S.A. Human Molecular Genetics. (2020)
Barnum C.E., Saai S.A., Patel S.D., Cheng C., Anand D., Xu X., Dash S., Siddam A.D., Glazewski L., Paglione E., Polson S., Chuma S., Mason R.W., Wei, S., Batish M., Fowler V.M., Lachke S.A. Human Molecular Genetics. (2020)
2018
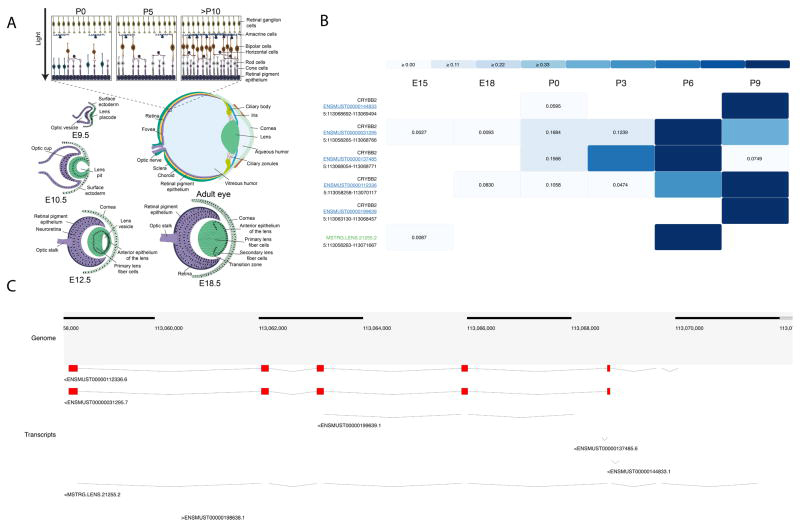
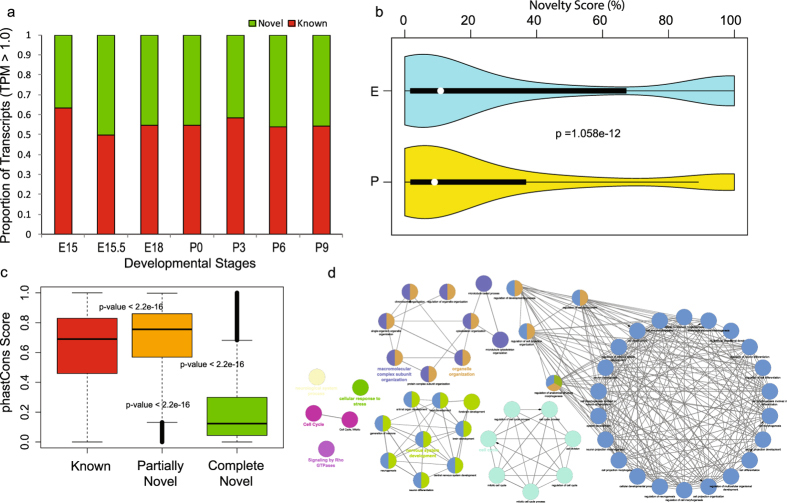
Express: A database of transcriptome profiles encompassing known and novel transcripts across multiple development stages in eye tissues.
Budak G., Dash S., Srivastava R., Lachke S.A., Janga S.C. (2018) Experimental Eye Research;168:57-66
Budak G., Dash S., Srivastava R., Lachke S.A., Janga S.C. (2018) Experimental Eye Research;168:57-66
2017
Transcriptome analysis of developing lens reveals abundance of novel transcripts and extensive splicing alterations.
Srivastava R., Budak G., Dash S., Lachke S.A., Janga S.C. (2017) Scientific Reports;7(1):115732.
Srivastava R., Budak G., Dash S., Lachke S.A., Janga S.C. (2017) Scientific Reports;7(1):115732.
2016
RNA Binding Proteins in Eye Development and Disease: Implication of Conserved RNA Granule Components.
Dash S., Siddam A.D., Barnum C.E., Janga S.C., Lachke S.A. (2016) WIREs RNA, 7(4):527-557.
Dash S., Siddam A.D., Barnum C.E., Janga S.C., Lachke S.A. (2016) WIREs RNA, 7(4):527-557.
2015
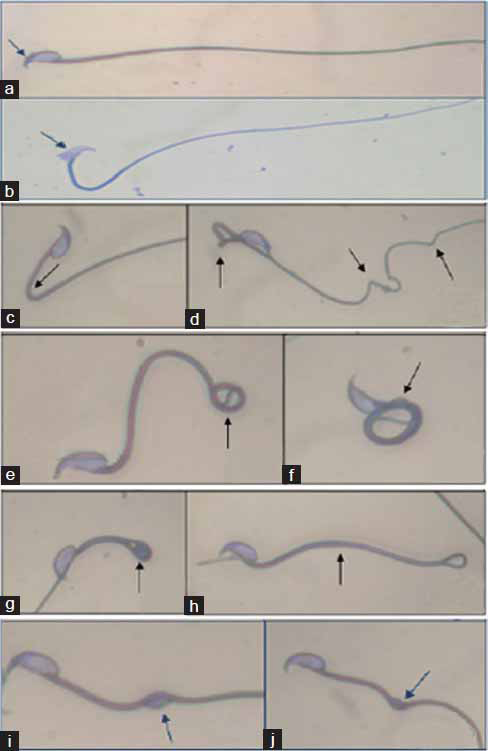
Anatase Titanium Dioxide Nanoparticles in Mice: Evidence for Induced Structural and Functional Sperm Defects after Short-, but not Long-, term Exposure.
Smith M.A., Michael S.R., Aravindan G. R., Dash S., Shah I., Galileo D.S., Martin-DeLeon P.A. (2015) Asian Journal of Andrology; 17(2):261-8.
Smith M.A., Michael S.R., Aravindan G. R., Dash S., Shah I., Galileo D.S., Martin-DeLeon P.A. (2015) Asian Journal of Andrology; 17(2):261-8.
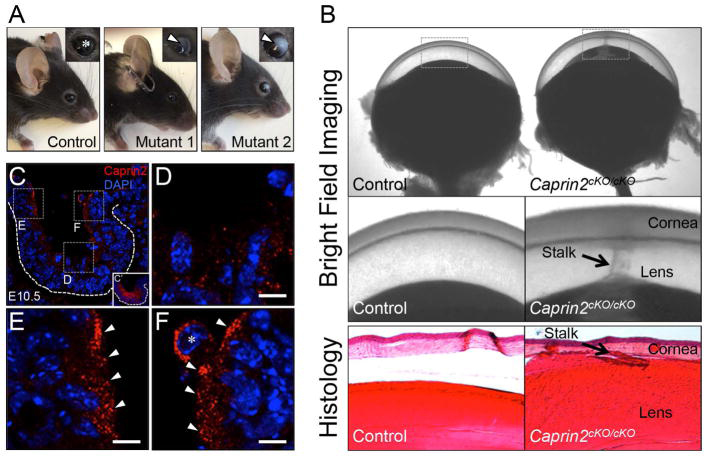
Deficiency of the RNA binding protein caprin2 causes lens defects and features of peters anomaly.
Dash S., Dang C.A., BeebeD.C., Lachke S.A. (2015) Development Dynamics;244(10):1313-27.
Dash S., Dang C.A., BeebeD.C., Lachke S.A. (2015) Development Dynamics;244(10):1313-27.
Compound mouse mutants of bZIP transcription factors Mafg and Mafk reveal a regulatory network of non-crystallin genes associated with cataract.
Agrawal S.A., Anand D., Siddam A.D., Kakrana A., Dash S., Scheiblin D.A., Dang C.A., Terrell A.M., Waters S.M., Singh A., Motohashi H., Yamamoto M., Lachke S.A. (2015) Human Genetics; 134(7):717-35.
Agrawal S.A., Anand D., Siddam A.D., Kakrana A., Dash S., Scheiblin D.A., Dang C.A., Terrell A.M., Waters S.M., Singh A., Motohashi H., Yamamoto M., Lachke S.A. (2015) Human Genetics; 134(7):717-35.
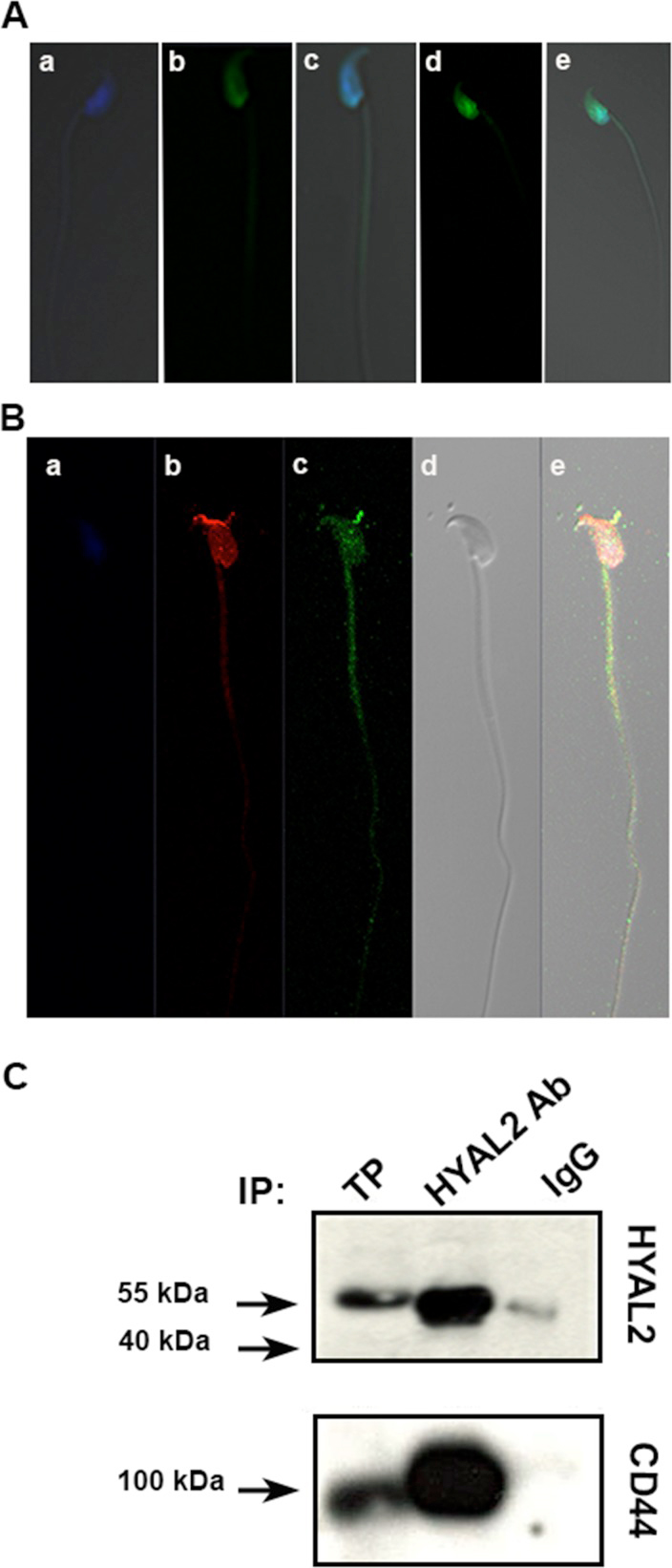
Hyaluronidase 2: A Novel Germ Cell Hyaluronidase with Epididymal Expression and Functional Roles in Mammalian Sperm.
Modelski M., Menlah G., Wang Y., Dash S., Wu K., Galileo D.S., and Martin-DeLeon P.A. (2014) Biology of Reproduction; 91(5):109 1-11.
Modelski M., Menlah G., Wang Y., Dash S., Wu K., Galileo D.S., and Martin-DeLeon P.A. (2014) Biology of Reproduction; 91(5):109 1-11.
Conference Talks and Posters
*Dash S., Bhatt S., Trainor P.A. Mediator complex subunit, Med23 is required in both neural crest cells and endothelial cells to regulate craniofacial development. Poster presented at 2021 North-East Society of Developmental Biology Regional Meeting, Online.
*Won First Place in Poster Competition
*Dash S., Bhatt S., Terrazas-Falcon K., Sandell, L.L., Trainor P.A. Mediator Complex Subunit, Med23 Regulates Craniofacial Development by Repressing Sox9 Expression. Poster presented at 2020 annual meeting of the Society for Craniofacial Genetics and Developmental Biology, Online.
*Won Third Place in Poster Competition
*Dash S., Trainor P.A. RNA Pol I subunits and associated protein Treacle, are required to maintain nucleolar structure. Short talk and poster presented at ASBMB Special Symposium Emerging Roles for the Nucleolus, October 24-27, 2019, Kansas City, MO, USA
*Dash S., Bhatt S., Terrazas-Falcon K., Trainor P.A. A novel role for the mediator complex protein Med23 in craniofacial development and in the pathogenesis of Pierre-Robin Sequence. Talk presented at 2019 annual meeting of the Society for Craniofacial Genetics and Developmental Biology, October 14-15, 2019, Houston, TX, USA
*Won Best Trainee Presentation Award
*Dash S., Brastorm M.K., Scott A.C., Slusarski D.C., Lachke S.A. (2017) Rbm24-mediated post-transcriptional gene regulation is necessary for vertebrate eye development. Paper presented at Rustbelt RNA Meeting, October 13-14, Indianapolis, IN, USA
*Dash S., Brastorm M., Slusarski D.C., Lachke S.A. (2017) Characterization of a New RNA-Binding Protein Rbm24 in Vertebrate Eye Development. Paper presented at ARVO Annual meeting, May 7-11, Baltimore, MD, USA
*Selected for Oral Presentation (top ~5% of >8000 abstracts)
*Borders N., Dash S., Lachke S.A. (2015) Functional characterization of a novel BTB/POZ domain zinc finger transcription factor ZBTB8B in mammalian lens development. Poster presentation at 18th Annual Undergraduate Research symposium in the Chemical and Biological Sciences, October 3, UMBC
*Won first place in Biological Sciences
I was involved in directly supervising Mr. Borders for his Undergraduate Senior Thesis research
*Dash S., Anand D., Lachke S.A. (2015) A novel BTB/POZ domain zinc finger transcription factor Zbtb8b functions in lens development. Presented at ARVO Annual meeting, May 3-7, Denver, CO, USA
*Selected for Oral Presentation (top ~5% of >8000 abstracts)
*Dash S., Dang C., Beebe D.C., Lachke S.A. (2014) Investigation of Caprin2 (RNG140 RNA granule protein 140) function in mouse lens. Poster presented at ARVO Annual meeting, May 4-8, Orlando, FL, USA
*Won MIT Outstanding Poster Award at ARVO(top 1 among ~200 abstracts considered)
*Dash S., Dang C., Beebe D.C., Lachke S.A. (2014) Investigation of Caprin2 function in lens fiber cell homeostasis. ePoster talk and poster presented at American Society for Cell Biology (ASCB) Annual meeting, December 6-10, Philadelphia, PA, USA
*Selected for ePoster talk (only 112 were selected from over 268 abstracts considered)
*Won First Place in Poster Competition
*Dash S., Bhatt S., Terrazas-Falcon K., Sandell, L.L., Trainor P.A. Mediator Complex Subunit, Med23 Regulates Craniofacial Development by Repressing Sox9 Expression. Poster presented at 2020 annual meeting of the Society for Craniofacial Genetics and Developmental Biology, Online.
*Won Third Place in Poster Competition
*Dash S., Trainor P.A. RNA Pol I subunits and associated protein Treacle, are required to maintain nucleolar structure. Short talk and poster presented at ASBMB Special Symposium Emerging Roles for the Nucleolus, October 24-27, 2019, Kansas City, MO, USA
*Dash S., Bhatt S., Terrazas-Falcon K., Trainor P.A. A novel role for the mediator complex protein Med23 in craniofacial development and in the pathogenesis of Pierre-Robin Sequence. Talk presented at 2019 annual meeting of the Society for Craniofacial Genetics and Developmental Biology, October 14-15, 2019, Houston, TX, USA
*Won Best Trainee Presentation Award
*Dash S., Brastorm M.K., Scott A.C., Slusarski D.C., Lachke S.A. (2017) Rbm24-mediated post-transcriptional gene regulation is necessary for vertebrate eye development. Paper presented at Rustbelt RNA Meeting, October 13-14, Indianapolis, IN, USA
*Dash S., Brastorm M., Slusarski D.C., Lachke S.A. (2017) Characterization of a New RNA-Binding Protein Rbm24 in Vertebrate Eye Development. Paper presented at ARVO Annual meeting, May 7-11, Baltimore, MD, USA
*Selected for Oral Presentation (top ~5% of >8000 abstracts)
*Borders N., Dash S., Lachke S.A. (2015) Functional characterization of a novel BTB/POZ domain zinc finger transcription factor ZBTB8B in mammalian lens development. Poster presentation at 18th Annual Undergraduate Research symposium in the Chemical and Biological Sciences, October 3, UMBC
*Won first place in Biological Sciences
I was involved in directly supervising Mr. Borders for his Undergraduate Senior Thesis research
*Dash S., Anand D., Lachke S.A. (2015) A novel BTB/POZ domain zinc finger transcription factor Zbtb8b functions in lens development. Presented at ARVO Annual meeting, May 3-7, Denver, CO, USA
*Selected for Oral Presentation (top ~5% of >8000 abstracts)
*Dash S., Dang C., Beebe D.C., Lachke S.A. (2014) Investigation of Caprin2 (RNG140 RNA granule protein 140) function in mouse lens. Poster presented at ARVO Annual meeting, May 4-8, Orlando, FL, USA
*Won MIT Outstanding Poster Award at ARVO(top 1 among ~200 abstracts considered)
*Dash S., Dang C., Beebe D.C., Lachke S.A. (2014) Investigation of Caprin2 function in lens fiber cell homeostasis. ePoster talk and poster presented at American Society for Cell Biology (ASCB) Annual meeting, December 6-10, Philadelphia, PA, USA
*Selected for ePoster talk (only 112 were selected from over 268 abstracts considered)
DEI Statement
As an immigrant woman of color, promoting diversity and inclusion in academia and sciences is extremely important to me. I have actively promoted diversity and inclusion by opening and participating in important discussions within my scientific field. My approach is to stay aware of the barriers that exist for under-represented groups, show appreciation for existing diversity so that everyone feels respected and valued, and take action to create opportunities to promote diversity and inclusion.