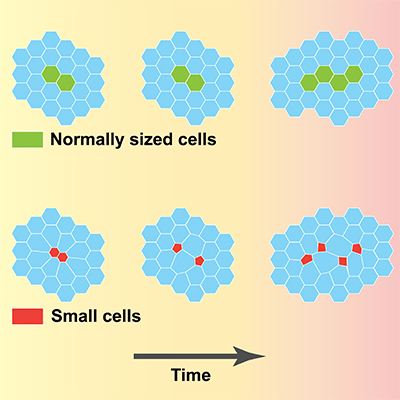
As epithelial tissues develop, groups of cells related by descent tend to associate in clonal populations rather than dispersing within the cell layer. While this is frequently assumed to be a result of differential adhesion, precise mechanisms controlling clonal cohesiveness remain unknown. Here we employ computational simulations to modulate epithelial cell size in silico and show that junctions between small cells frequently collapse, resulting in clone-cell dispersal among larger neighbors. Consistent with similar dynamics in vivo, we further demonstrate that mosaic disruption of Drosophila Tor generates small cells and results in aberrant clone dispersal in developing wing disc epithelia. We propose a geometric basis for this phenomenon, supported in part by the observation that soap-foam cells exhibit similar size-dependent junctional rearrangements.Combined, these results establish a link between cell-size pleomorphism and the control of epithelial cell packing, with potential implications for understanding tumor cell dispersal in human disease.
From:
Cell size pleomorphism drives aberrant clone dispersal in proliferating epithelia.
Ramanathan S. P., Krajnc, M., Gibson, M.C.
Developmental Cell (2019) 51: 49-61.
PubMed
View PDF